myvanwygibson
November 29, 2015 8:33 pm
Adhesion to Plastics
Surface tension actually has its own unit of measurement called dynes or dynes per centimeter, expressed dynes/cm. Clean, well prepared metal surfaces have high surface energies in the order of 400 to 1800 dynes/cm (Aluminum ~500, Copper ~ 1300, Nickel ~ 1800). They are wetted easily by acrylic polymers that have relatively low surface tension in the order of 40 to 50 dynes/cm. Wetting of plastic surfaces, on the other hand, is much more complex than wetting metal surfaces. Plastics being non-porous, non-polar or low-polar, hydrophobic, and being low surface energy materials are difficult to adhere to not just for artists, but also within the automotive, electronics and medical industries.
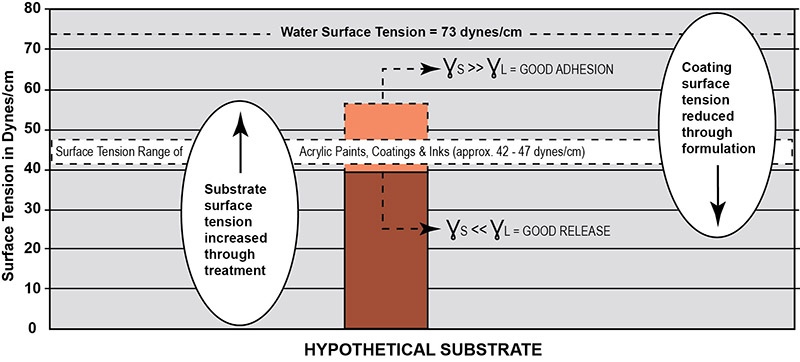
Plastic surfaces and paints are both polymeric materials and thus have similar surface tensions. There is considerable surface tension closeness or overlap within the narrow band between the plastic substrates (20 to 50 dynes/cm) and the waterborne acrylic dispersion polymers (39 to 47 dynes/cm) as shown in Figure 2. Adhesion between paint and the substrate is dependent on many factors such as differential surface tension, differential expansion to hot and cold, differential modulus (a measure of stiffness), shrinkage during drying/cure, the effects of solvents in the formulation, chemical structure, etc. Yet the most important factor determining adequate adhesion is the differential surface tension. The absence of a large difference between the critical surface tension of the paint and plastic substrates often results in poor surface adhesion or even complete failure. As a general rule, acceptable bonding adhesion is achieved when the surface energy of a substrate (measured in dynes/cm) is approximately 10 dynes/cm greater than the surface tension of the liquid. In this situation, the liquid is said to "wet out" or adhere to the surface. For this to happen, either the surface tension of the substrate or the paint must break loose of the logjam in Figure 2. An illustration of the surface energy of a hypothetical plastic substrate for good adhesion to waterborne acrylic is presented in Figure 3. As shown here, the higher the surface energy of the solid substrate relative to the surface tension of a liquid, the better its wettability will be, and the smaller the contact angle.
Adhesion strength is generally determined by the properties of a base material and its interface. Optimizing adhesion strength can be accomplished by modifying these interfaces physically and chemically. In formulating waterborne acrylic paints, the use of surface acting modifiers (surfactants) is essential in driving down the surface tension of the liquid paints. These materials can take the acrylic dispersion polymer's surface tension down between 26 to 31 dynes, allowing wetting out of many difficult to coat surfaces.